Description
Bicosome® FS
| Subject | Description |
|---|---|
| Appearance | Yellowish Liquid |
| pH | 3.0 – 6.0 |
| Composition | – AQUA UP TO 100% – LECITHIN 5-10% – HYDROGENATED PHOSPHATIDYLCHOLINE 1-10% – LYSOLECITHIN 1 – 10% |
| Applications | Cosmetic products |
| Suggested Concentration | 1 – 3% |
| Function | Skin firming |
| INCI Name | Water, Lecithin, Hydrogenated Phosphatidylcholine, Lysolecithin, Sodium Benzonate, Citric Acid, Potassium Sorbate, Phytic Acid, Tocophenol, Beta-Sitosterol,Squalene |
| Storage conditions | Store the product tightly closed in a dry and ventilated place, at not more than 8ºC. Protect from sunlight. The results of stability studies allow short periods of transport at 25ºC. Stirring the full content of the container before use or fractioning it is recommended to ensure adequate homogeneity. Use soon after opening the package. |
| Shelf-Life | 24 months from manufacturing date |
Bicosome® FS stands out for its innovative Bicosome® Technology, which mimics the natural lipid structure of human skin. It features a unique dual structure composed of an outer lipidic vesicle that merges with the outermost skin layer, and internal discoidal structures that penetrate through intercellular spaces into the middle region of the stratum corneum.
ATR-FTIR spectroscopy using deuterium-labeled lipids demonstrated that Bicosome® FS disperses into the mid-stratum corneum. Additionally, GISAXS (Grazing Incidence Small Angle X-Ray Scattering) analysis showed that Bicosome® FS enhances lipid organization within the skin, creating new lamellar structures that integrate into the skin’s lipid matrix—mimicking the function of the basal epidermal layers responsible for forming the skin’s lipid architecture.
The product has passed comprehensive safety testing, confirming it to be non-irritant, non-phototoxic, and non-mutagenic. It is certified Vegan, Non-GMO, China NMPA compliant, and is not classified as a nanomaterial.
Recommended usage level: 1–3% in all types of skincare formulations.
Mechanism of Action and Composition
Research Study by BICOSOME, S.L.
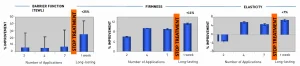
Short-Term & Long-Lasting Effects Clinical Study
Method:
A study was conducted on 20 female volunteers aged 25–38 over a period of 16 days. A formula containing 2% Bicosome® FS and a control formula (without Bicosome® FS) were applied to the inner forearm once daily, for a total of 7 applications over 9 days. The subjects then stopped using the products, and results were monitored for an additional week.
Skin Barrier Function (TEWL), elasticity, and firmness were measured using a Tewameter TM 300 and a Cutometer SEM 575 during the application period (short-term efficacy) and again one week after discontinuation (long-lasting effect).
Research Study
Reference : Coderch, L., et al. “Ceramides and skin function: role in barrier properties.” American Journal of Clinical Dermatology, 2003, 4(2): 107-129
Product Safety and Warranty Information
- The product is manufactured through a certified process by leading organizations and has certification for industry standards that can be referenced.
-
The product is safe and can be used to support the registration of a cosmetic product.
- The product is a raw material for industrial use, not a finished product. Users must have the knowledge and appropriate tools for using the raw material.
- The product is warranted according to the specifications, please refer to the terms and conditions for details.